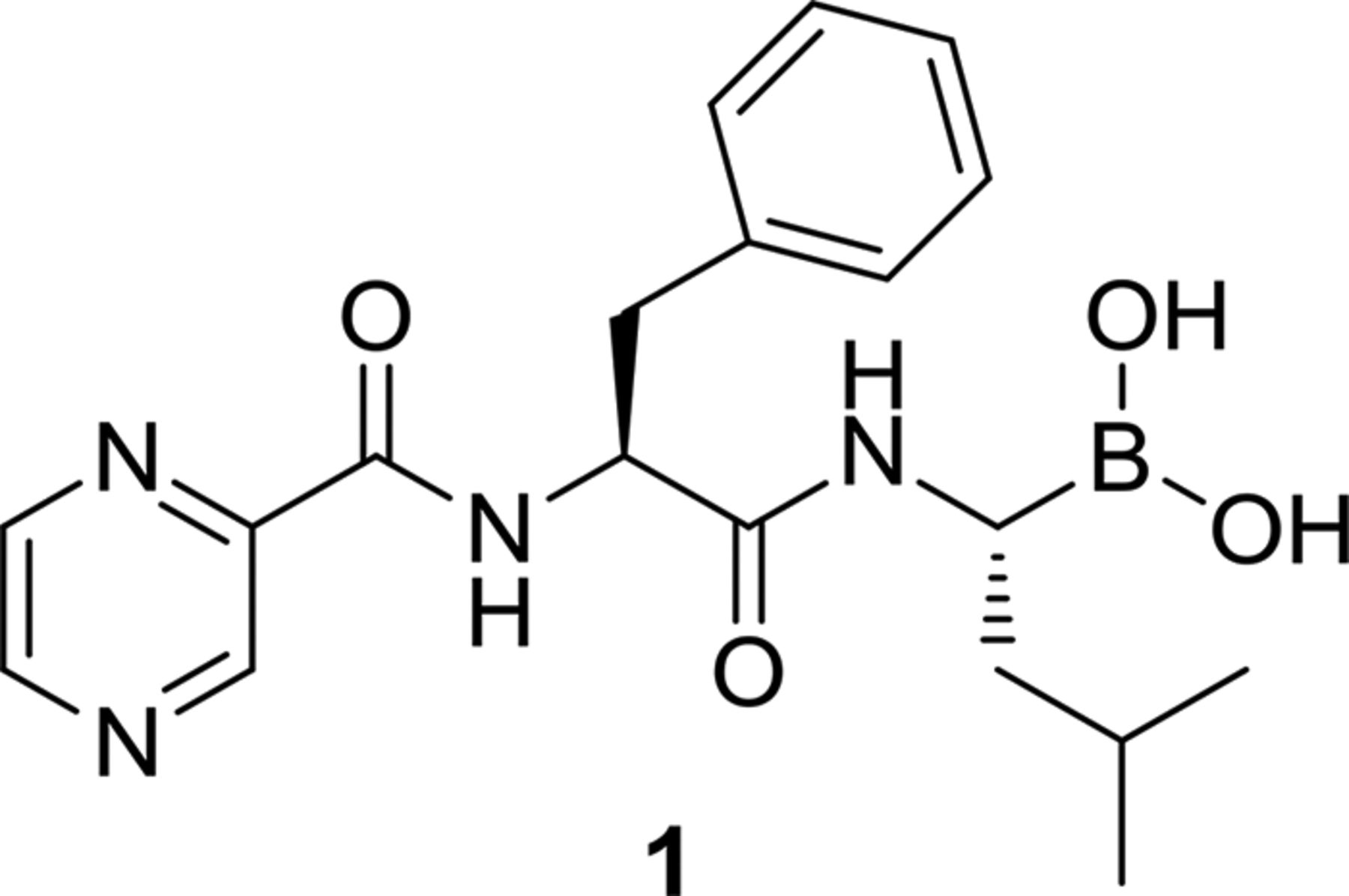
The human proteasome inhibitor Bortezomib was identified as a potent inhibitor of mycobacterial ClpP in a target mechanism-based whole cell screen. The initial phase of lead optimization aimed to introduce selectivity of the series for the mycobacterial enzyme. A series of novel analogues have been prepared as mycobacterial caseinolytic protease ClpP1P2 inhibitors. Compounds were characterized in a ClpP1P2 target-based cell reporter assay, confirming protease inhibition and bacterial-cell penetration. This demonstrated that selective, potent inhibitors of ClpP1P2 are possible and could pave the way for further studies, leading to new treatment options for drug-resistant M. tuberculosis.
Wikipedia: Bortezomib is an N-protected dipeptide that can be written as Pyz-Phe-boroLeu, which stands for pyrazinoic acid, phenylalanine and leucine with a boronic acid instead of a carboxylic acid.
Compound
Bortezomib
SPRINT TB (National University of Singapore)
Description
Related Links
Developer Associations