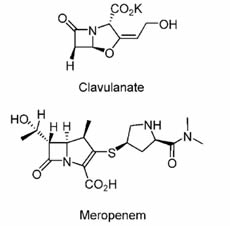
Beta-lactam antibiotics are ineffective against Mycobacterium tuberculosis, being rapidly hydrolyzed by the chromosomally encoded blaC beta-lactamase. The carbapenem class of b-lactams are very poor substrates for BlaC. Meropenem and clavulanate are US FDA–approved drugs and could potentially be used to treat patients with currently untreatable disease.
A clinical trial was developed to evaluate the early bactericidal activity (EBA), safety, tolerability and pharmacokinetics of meropenem administered intravenously three times a day, plus amoxycillin/CA administered orally three times a day; and of faropenem administered orally three times a day, plus amoxycillin/CA administered orally three times a day. Drugs were administered for 14 consecutive days, in adult participants with newly diagnosed, smear positive pulmonary tuberculosis, in order to help establish proof-of-concept for carbapenem antibiotics as antituberculosis agents and to select the appropriate agent and route of administration for later stage clinical development.
Compound
Meropenem / Clavulanate
Description
Related Links
- Hugonnet et al. 2009.Meropenem clavulanate effective against XDR TB.Science.323(5918):1215-8
- Dhar et al. 2015. Rapid cytolysis of Mtb by Faropenem, an orally available beta lactam antibiotic.AAC.59(2):1308-19.
- Hugonnet, Blanchard.2007. Irreversible inhibition of the Mycobacterium tuberculosis beta-lactamase by clavulanate. Biochemistry.46(43):11998-2004
- Holzgrabe. 2009. Meropenem-clavulanate: a new strategy for the treatment of tuberculosis? ChemMedChem. 4(7):1051-3
Trial Associations