Using structure-based design, a new semisynthetic series of spectinomycin analogs with selective ribosomal inhibition and narrow-spectrum antitubercular activity had been generated. In multiple murine infection models, these spectinamides were well tolerated, significantly reduced lung mycobacterial burden and increased survival. In vitro studies demonstrated a lack of cross resistance with existing tuberculosis therapeutics, activity against multidrug-resistant (MDR) and extensively drug-resistant tuberculosis and an excellent pharmacological profile. Key to their potent antitubercular properties is the structural modification to evade the Rv1258c efflux pump, which is upregulated in MDR strains and is implicated in macrophage-induced drug tolerance. The antitubercular efficacy of spectinamides demonstrates that synthetic modifications to classical antibiotics can overcome the challenge of intrinsic efflux pump-mediated resistance and expands opportunities for target-based tuberculosis drug discovery.
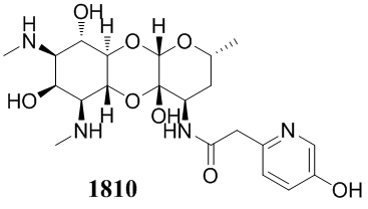
Lee 1810 was recently selected as the preclinical spectinamide candidate due to its excellent safety profile (Maximum Tolerated Dose of 500 mg/kg by IV infusion) and its efficacy in multiple murine models of TB infection. In addition, it maintains the activity against MDR and XDR strains of TB, avoidance of efflux by the Rv1258c pump, excellent stability, low protein binding and pharmacokinetic parameters observed for Lee 1599, a previous lead candidate. Finally, Lee 1810 is synergistic with Rifampicin-Pyrazinamide combinations in murine models of TB infection (PMID:27999020). Preclinical studies on Lee 1810 began in early 2017.