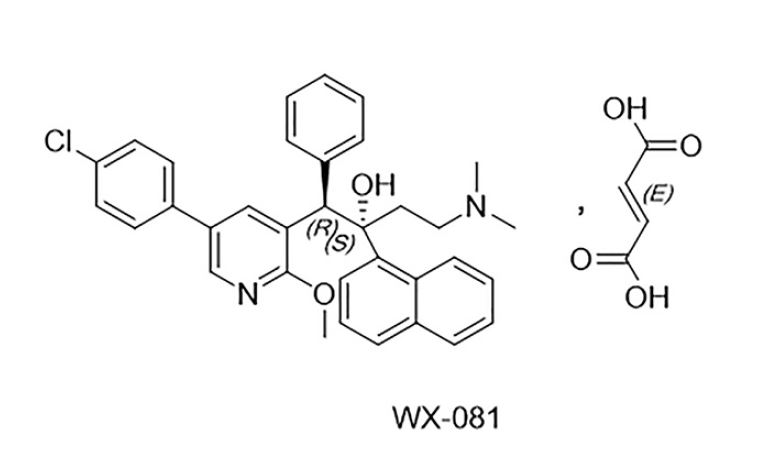
2022 Publication. Huang, Luo, Wang, Wan, Franzblau, Ding. Discovery and preclinical profile of sudapyridine (WX-081), a novel anti-tuberculosis agent. Bioorganic & Medicinal Chemistry Letters. Volume 71, 1 September 2022, 128824
Abstract: Multidrug resistant tuberculosis (MDR-TB) remains a major human health challenge. Bedaquiline was approved in 2012 by the US FDA, and listed by WHO as a treatment for multidrug-resistant tuberculosis (MDR-TB) in 2018. However, the side effects of bedaquiline including the risk of unexplained mortality, QTc prolongation and hepatotoxicity limit its wide clinical use. Based on bedaquiline, we describe herein discovery and development of a novel diarylpyridine series, which led to identification of WX-081 (sudapyridine). It displayed excellent anti-mycobacterial activity against M. tuberculosis H37Rv in vitro and in vivo and low cytotoxicity; additionally WX-081 had excellent pharmcokinetic parameters in animals, better lung exposure and lower QTc prolongation potential compared to bedaquiline. WX-081 is currently under clinical phase II development (NCT04608955). Corresponding author: Charles_Ding@wuxiapptec.com
2022 Publication. Yao et al. Sudapyridine (WX-081), a Novel Compound against Mycobacterium tuberculosis. Microbiol Spectrum. 10.1. e02477-21. PMCID: PMC8849072
From the abstract: Investigators in China report " a new analog of bedaquiline, sudapyridine (WX-081), was selected as a potential new antituberculosis candidate to move into clinical trials. Here, we evaluated WX-081's overall preclinical profile, including efficacy, pharmacokinetics, and toxicology. The in vitro activity of WX-081 against drug-sensitive and drug-resistant tuberculosis was comparable to that of BDQ, and there was comparable efficacy between WX-081 and BDQ in both acute and chronic mouse tuberculosis models using low-dose aerosol infection. Moreover, WX-081 improved pharmacokinetic parameters and, more importantly, had no adverse effects on blood pressure, heart rate, or qualitative ECG parameters from nonclinical toxicology studies. WX-081 is under investigation in a phase 2 study in patients." And from the Shanghai Jiatan Biotech Ltd., a subsidiary of Guangzhou JOYO Pharma Ltd., Shanghai, China the following updates were provided October 2021:
1. Full Phase I including SAD, Food Effect and MAD completed and CSR wrapped up.
2. Phase II trial including EBA and extension for primary efficacy is near Last Patient-In, and expect to complete in the 1st half of 2022.
3. Now heading to Phase III preparation including Protocol Design and Site Selection, and plan to start Phase III in the 1Q of 2022 in China.
4. Long-term nonclinical tox study including 26 weeks in rat and 39 weeks in dog completed.