TBAJ-587 began Phase 1 clinical trial October 2020.
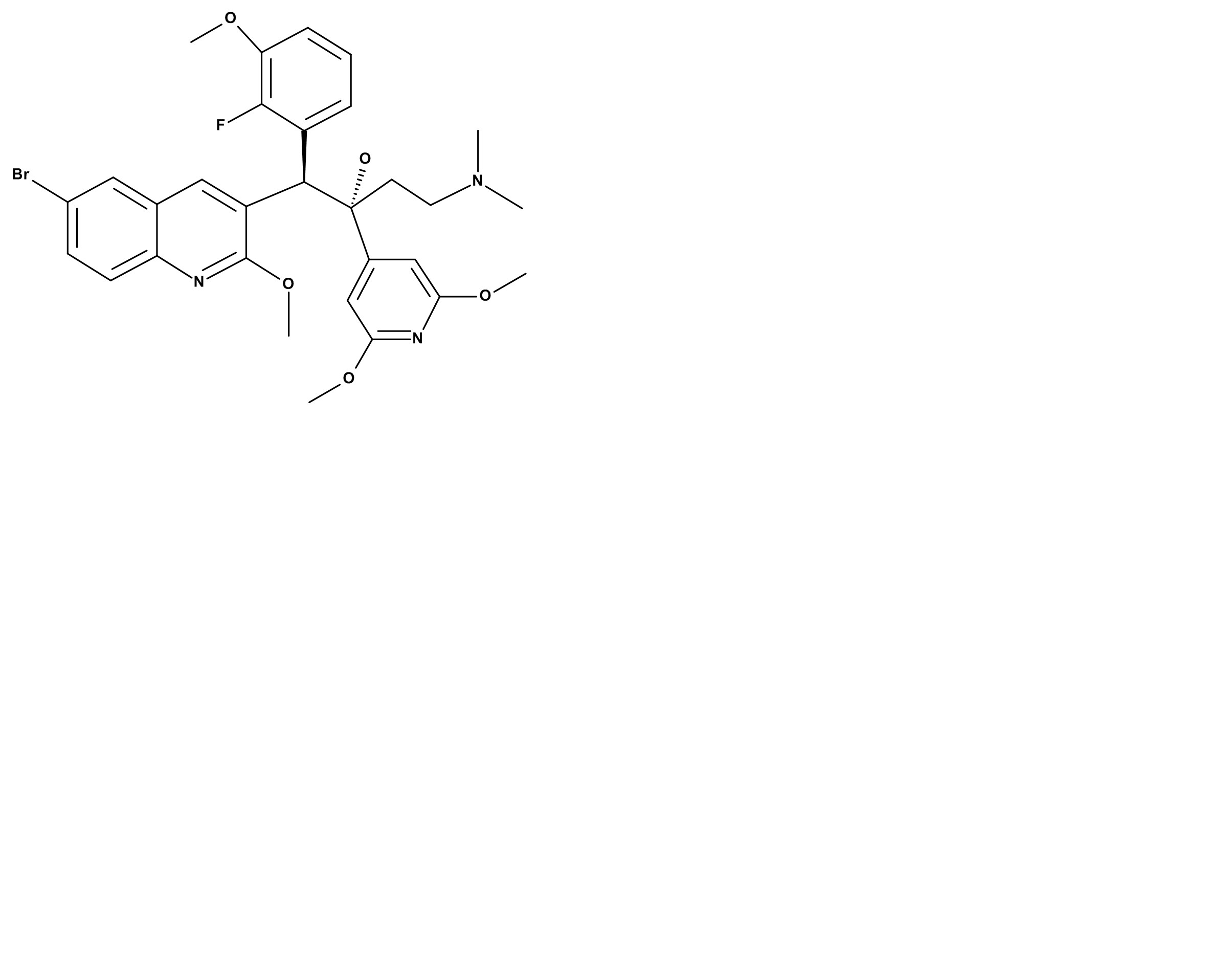
Bedaquiline represents both a novel chemical class - diarylquinolines (DARQs) - as well as a novel M. tuberculosis-selective mechanism of action (direct inhibition of M.tb ATP synthase). TBAJ-587 was identified following a next-generation diarylquinoline lead optimization project that sought to identify novel members of this chemical series, which maintain bedaquiline’s impressive anti-TB activities and possess improved properties. TBAJ-587 has more potent activity against M. tuberculosis and better efficacy in animal models of TB. Preliminary studies suggest it has a better safety and pharmacokinetic profile than bedaquiline.
Compound
TBAJ-587, Diarylquinoline
TB Alliance, ERA4TB (European Regimen Accelerator for Tuberculosis), University of Auckland, Merck & Co., Inc.
Description
Related Links
Developer Associations
ERA4TB (European Regimen Accelerator for Tuberculosis)
Downloads