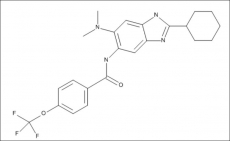
Trisubstituted benzimidazoles have demonstrated potency against Gram-positive and Gram-negative pathogens. Previously, a library of novel trisubstituted benzimidazoles was constructed for high throughput screening, and compounds were identified that exhibited potency against M. tuberculosis H37Rv and clinical isolates, and were not toxic to Vero cells. A new series of 2-cyclohexyl-5-acylamino-6-N, N-dimethylaminobenzimidazoles derivatives has been developed based on SAR studies. Screening identified compounds with potency against M. tuberculosis. A lead compound from this series, SB-P17G-A20, was discovered to have an MIC of 0.16 µg/mL and demonstrated efficacy in the TB murine acute model of infection based on the reduction of bacterial load in the lungs and spleen by 1.73 ± 0.24 Log10 CFU and 2.68 ± Log10 CFU, respectively, when delivered at 50 mg/kg by intraperitoneal injection (IP) twice daily (bid). The activity of SB-P17G-A20 was determined to be concentration dependent and to have excellent stability in mouse and human plasma, and liver microsomes. Together, these studies demonstrate that SB-P17G-A20 has potency against M. tuberculosis clinical strains with varying susceptibility and efficacy in animal models of infection, and that trisubstituted benzimidazoles continue to be a platform for the development of novel inhibitors with efficacy. In mouse and human plasma, and liver microsomes, SB-P17G-A20 was concentration dependent and had excellent stability (PLoS One, 15 Apr 2014, 9, PMID:24736743, http://www.ncbi.nlm.nih.gov/pubmed/24736743).
Compound
Trisubstituted benzimidazoles
Description